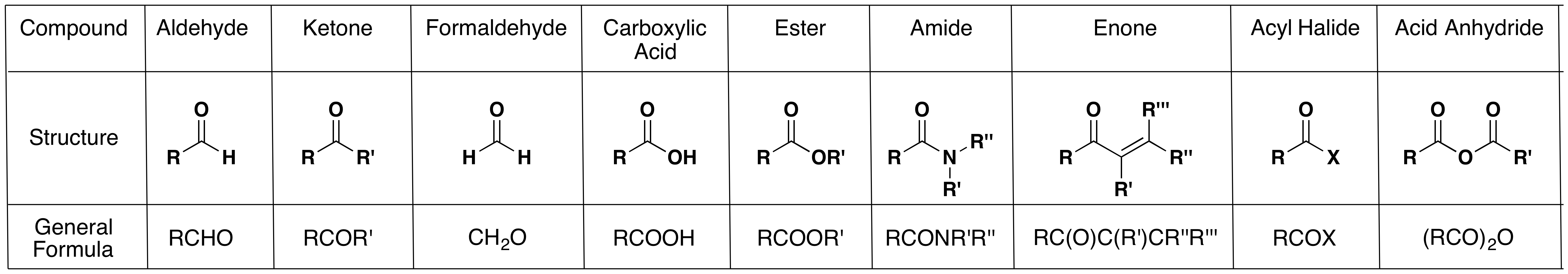
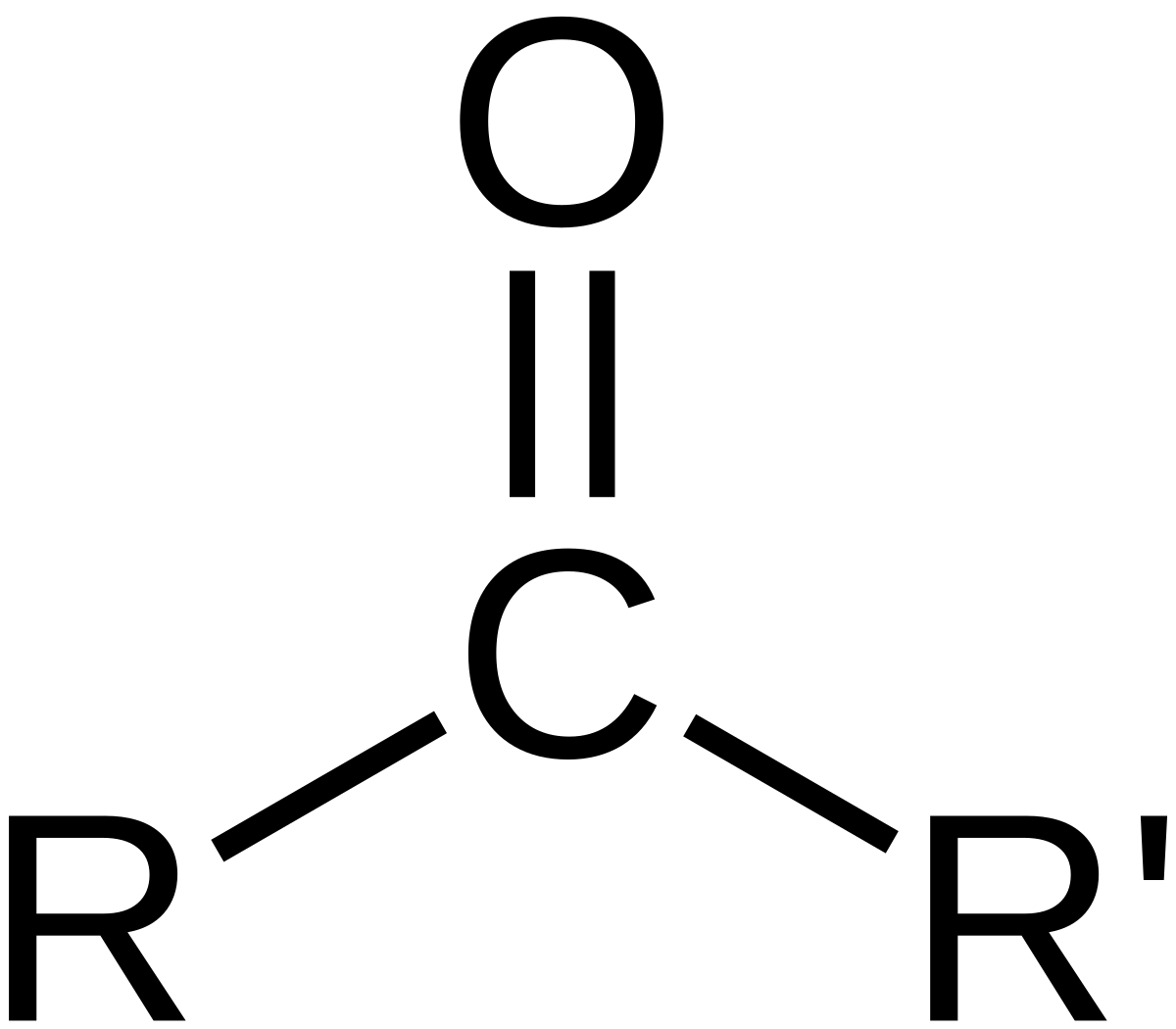
In chemistry, a ketone (alkanone) / ˈ k iː t oʊ n / is an organic compound with the structure RC(=O)R’, where R and R’ can be a variety of carbon-containing substituents.Ketones and aldehydes are simple compounds that contain a carbonyl group (a carbon-oxygen double bond).

Carbonyl group: Carbonyl group,, in organic chemistry, a divalent chemical unit consisting of a carbon (C) and an oxygen (O) atom connected by a double bond. The group is a constituent of carboxylic acids, esters, anhydrides, acyl halides, amides, and quinones, and it is the characteristic functional group
Ketone sind chemische Verbindungen, die als funktionelle Gruppe eine nicht endständige Carbonylgruppe (>C=O) enthalten. Eine Ketogruppe [C–C(O)–C] enthält drei Kohlenstoffatome.

Carbonyl Chemistry: Survey of Reactions and Mechanisms: When dealing with Carbonyls, we consider two general mechanism types: Carbonyls Have X as a Leaving Group Don’t have X as a Leaving Group
Ketone: Ketone, any of a class of organic compounds characterized by the presence of a carbonyl group in which the carbon atom is covalently bonded to an oxygen atom.
An enone, also called an α,β-unsaturated carbonyl, is a type of organic compound consisting of an alkene conjugated to a ketone. The simplest enone is methyl vinyl ketone (butenone) or CH 2 =CHCOCH 3.

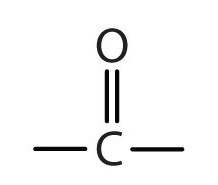
Ketone definition, any of a class of organic compounds containing a carbonyl group, CO, attached to two alkyl groups, as CH 3 COCH 3 or CH 3 COC 2 H 5 . See more.
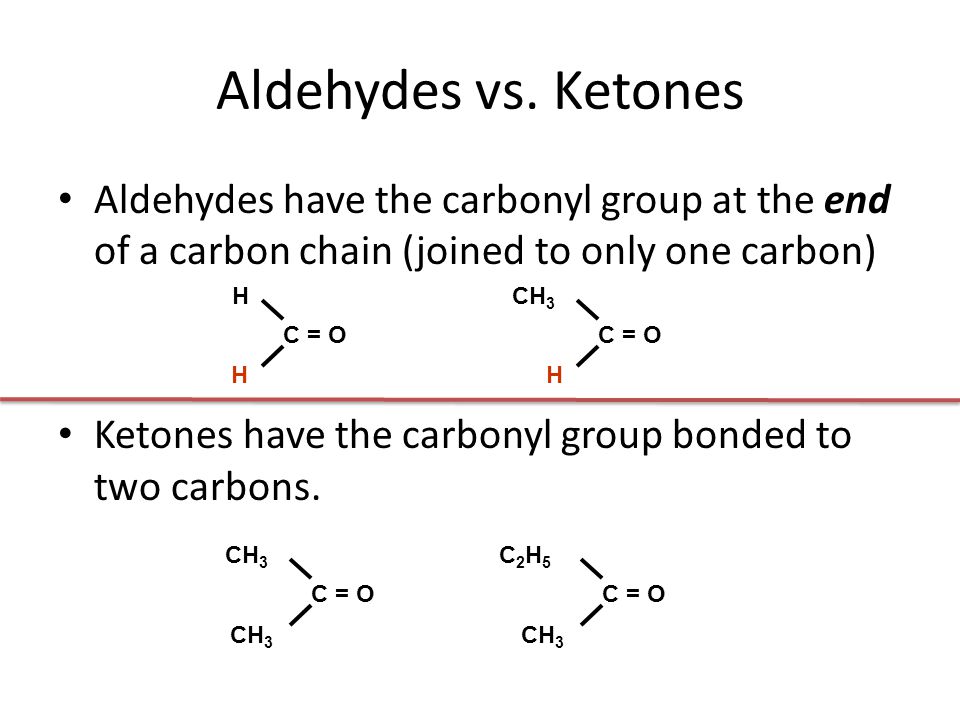
Aldehydes and Ketones 1. Nomenclature of Aldehydes and Ketones. Aldehydes and ketones are organic compounds which incorporate a carbonyl functional group, C=O.The carbon atom of this group has two remaining bonds that may be occupied by hydrogen or alkyl or aryl substituents.

: any of a class of organic compounds (such as acetone) characterized by a carbonyl group attached to two carbon atoms —
1 Overview of Carbonyl Compounds. 1. Kinds of Carbonyl Compounds. a) Aldehydes and ketones – RCOH and R 2CO.No leaving group attached to carbonyl …